Why Does Toxicological Risk Assessment Matter?
ISO 10993-1 requires a risk-based biological evaluation. Authorities want to see how chemical findings translate into real‑world exposure and patient safety.
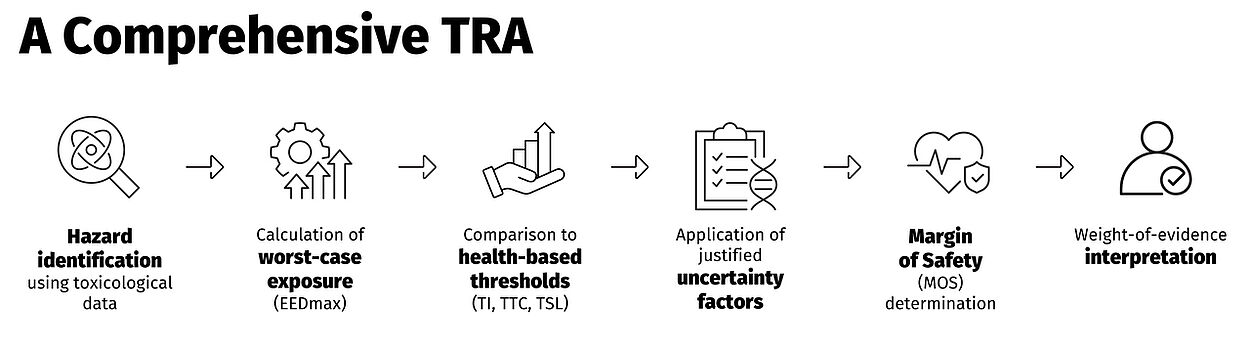
TRA answers the key questions:
- What substances were found?
- At what levels?
- How much would a patient be exposed to?
- How does this compare to toxicological thresholds?
TRA also supports:
- Root‑cause analysis when biological tests show unexpected results
- Informed decision-making early in development
- Continuous risk management throughout the product lifecycle